Exo Space Blog
Work, projects, and basically anything else I do in class
(Not just about space)
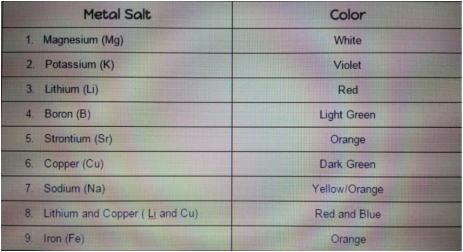 If I were to be given an unknown element, I would be able to use this information to figure out what element it is. I would burn it and let it rest so it can produce its color, then I would see if it matches with another color on the chart. With that I would be able to see what element it is. For this experiment we used a stance that held the fuel and the fuel tube, ten we used a spark to ignite the fire. Since the stance was Iron (Fe) so the flame was orange. Then Andrew got a bit of magnesium (Mg) and put it in the flame. As soon as it started to light up he lifted it away from the flame and it shined a bright white. It almost looked like the Lumos spell from harry potter. Then he dipped the small pipe in cold water to cool. We repeated this for all the metals and he let some people do it on their own. One interesting thing that happened was when Darian put in Lithium (Li) a bit of it fell into the flame and made it look red from then on. I really liked the small intro we had in the beginning because it gave us a better understanding of what was going on. It let me know why the light changes color and why it is different for different metals. All the metals looked cool in the flame, but my favorite had to be the Mg. I really liked this small experiment and hope to do more just like it. In the atoms of each element they have unoccupied energy levels. When energy is added to the atoms the electrons move up towards those levels, then once the energy is removed the electrons move back down and release there own spectrum of light. Each element has a different color because the atoms have different electrons and energy levels. Compared to other activities we have done in the classroom, this one was a bit more exciting. Unlike others where we had to see something that was exactly the same, this one was different every time and it amazed us and even the people around us that weren't doing the experiment. Overall I think it's fair to say that everyone enjoyed this experiment and we all learned a lot from it. I think this activity was fine the way it was, but if I really had to change or add something I would probably have more room so everyone can be spaced out for safety precautions and to let others see. One thing I took from this experiment was to always keep a cup of water close when working with hot/flammable objects. Also I learned that burning some metal salts can be really coo and at the same time very interesting so the next time I do a lab I should not push the research aside.
0 Comments
Leave a Reply. |
Jennifer R.Grade: 10th Archives
November 2015
Categories |
 RSS Feed
RSS Feed
