Exo Space Blog
Work, projects, and basically anything else I do in class
(Not just about space)
|
For this project we had to make a poster showing the lab safety procedure we were given. Mine was "Only the instructor, invited guest, and students involved in the project are allowed in the lab." Overall it was a pretty fun project and a great way to start the year.
0 Comments
Question: 1. How does the whipping cream form soft peaks; and how does the ice cream keep its flavor once frozen? 2. How does the chocolate bowl keep its shape once the balloon has been popped? Things I will Use: 1. Whipping Cream 8. Mixing Bowl 2. Condensed Milk 9. Hand Mixer 3. Mint Leaves or Mint Flavoring 10. Spatula 4. Chocolate Chips 11. Tray 5. Food Coloring (Green) 12. Parchment Paper 6. Chocolate Melts 13. Metal Bowl 7. Water Balloons 12. Teaspoon Steps: 1. Place whipping cream and condensed milk in fridge for 24 hours. 2. In the mixing bowl whip the whipping cream until soft peaks form. 3. Add condensed milk and whip again to combine ( this is the ice cream base). 4. Add some Mint Extract and combine with ice cream base. 5. Mix in green food coloring for color 6. Pour in some chocolate chips and fold mixture. 7. Once the desired flavor and appearance is achieved, place in freezer for 2 hours. 8. Blow up balloons and tie. 9. Wash balloons and dry. 10. Line a tray with parchment paper. 11. Melt chocolate melts in an oven safe bowl over a pot of boiling water. 12. With a spoon make small chocolate fill circles on the tray spreaded apart (base of the chocolate bowls). 13. Coat the bottom of each balloon with a thick layer of chocolate and place on top of each circle. 14. Place try in fridge until chocolate hardens. 15. Take out ice cream and chocolate bowls from the fridge/ freezer. 16. Pop each balloon and remove from chocolate. 17. Remove chocolate bowls from tray and place on a plate. 18. Scoop some ice cream and place in bowls. 19. Add desired toppings and enjoy. Expectation: 1. Whipping cream will form soft peaks and combine well with the condensed milk. 2. Mint leaves will add flavor to the ice cream. 3. Once frozen, Ice Cream will maintain flavor. 4. Chocolate will not drip off from balloon and will set in the shape of a bowl. 5. Chocolate won't crumble once balloon is popped. 6. Chocolate won't melt at room temperature and will be able to hold ice cream. 7. It will taste good. Post-Lab: Looking Back: Lot's of things happened, good and bad. I feel that the final product was okay, but it wasn't what I was going for.The ice cream was very delicious and maintained it's flavor even when frozen. It had the right amount of mint and chocolate chips, even the color was perfect. The chocolate bowls did form the shape of a bowl, and kept it even when the balloon popped. Even though the bowls came out, I couldn't really use them. The first problem was that every time I tried to put chocolate on the balloons, they would pop. I had to coat about twelve to get six come out. Then once I got them all and froze them, they were not great. The balloon popped and would stay stuck to the chocolate bowl. I could not take it off because the bowl would melt at my touch. In the end i could not use them as bowls. Looking Beyond: I think the way the ice cream froze and maintained it's flavor was interesting. I believe this happens because when the ice cream freezes, it does not lose its flavor, it locks it in. So one you take it out from the fridge and it starts to melt in your mouth, the favor are released and you can taste it. Still I do think some of the flavor goes away in the freezer, and that is why you should add more flavor than it needs. For the bowls I think the fridge locked in place all the molecules of the chocolate, but since it was chocolate chips instead of chocolate melts, it melted faster. Looking Inward: For the ice cream I did get desired results. I think this is because I kept looking back at my research for every step and I had my sister there to taste things and tell me what it's missing. The chocolate bowls were a bit more difficult. I should have used chocolate melts and should have waited a bit longer for the chocolate to cool. Because of this I did not get the result I wanted and ended up with a messy kitchen with chocolate splattered everywhere. Looking Outward: I was working on my own for this project because I feel more comfortable that way. I feel since I was doing all the work it was a bit too much having to do two things at a time, and getting all the supplies on my own. But I still learned a lot from it and I had fun doing it, even if I was on my own. Looking Forward: I think that next time I will do a few things differently. First, I would get the right chocolate to make the perfect bowls that do not melt to the touch. Second, I would make sure to do everything carefully and unrushed to limit my mistakes, like having the balloons pop. Lastly, I would do this with time so I am not worrying about this in the end. Pre-Lab Write Up:
Question: Why does the nailpolish sit on top of the water instead of sinking; and then sticks to the pot. Things I Will Use: 1. Nail polish in different colors (as many desired). 2. Plant Pot 3. Water 4. Bucket (tall and wide enough for pot to go in with ease) Steps: 1. Fill a bucket 3/4 of the way with water. 2. Pour each nail polish color into the bucket one at a time. 3. Dip the pot into the water making sure all sides touch the nail polish. 4. Leave to dry. Expectation: 1. The water and the nail polish will not combine. 2. The different nail polish colors will not combine with each other. 3. The nail polish will stick to the pot and will look like splattered paint. 4. One the pot dries the nail polish will still be in tact. Post-Lab: Looking Back: What I hoped would happened, did actually occur. The nail polish did not sink nor combined with the water and once the pot was removed from the water it looked colorful, but a bit messy. The water did not combine with the water which was good; and the different nail polish did not combine with each other. I was very worried that the nail polish I got would have a different effect then what I researched, but thankfully it acted the same. The main thing that went wrong as the fact that when the pot was removed from the water,It had blobs of paint dripping down. I realized that my pouring technique was the problem. When I poured the nail polish in the water I would shake the bottle causing blobs to form in it. So when I placed the pot the extra paint would run down the sides. If I were to do this lab again I would tr to let the nail polish run on its own without shaking the bottle. Looking Beyond: The density of the nail polish is different from the water, so when you pour it in, it just sits on the top. It doesn't combine either unless you mix it. Then since it just sits on the top, once you dip the pot, it sticks to it because it is like paint and the pot removes it from the water. Looking Inward: The nail polish did not combine with the water which is exactly what I wanted, but the final product didn't look so great. There were blobs of paint on the pot, and paint was still running down. I think the result would have come out better if I would have done more research. Looking Outward: I worked on my own for this project because it was a pretty simple project. I believe that since this was a small experiment, I did not really think much of it and thought it was as simple as just pouring and dipping. Getting the materials was not much of a problem, but getting them on time was. Looking Forward: If I were to do this experiment again I would change the way I poured the nail polish. I was shaking the bottle while pouring, which is why blobs were formed in the water and ran down the pot. I think with this in mind, next time they will come out better. The quote I chose is the one stated above. I chose this quote because it really struck me. To be honest throughout most of the video I was trying to come up with the perfect answer; but with every answer I just had more questions. I think it is one of those quotes that could be argued/discussed, but never really answered or defined. To me this quote means, or is trying to say that, would you spend your life knowing all the possibilities out there, but at the same time having the fear of one of them happening and creating a devastating disaster. Or would you live not know what is out there in the world and what could happen, and continue life being clueless of what is going on. After hearing this quote my head filled up with thoughts and questions like what if I end up not learning and knowing everything I can, will my whole life just be a waste. Or what if I fear what I know and end up fearing living as much as I fear death. Will I be able to live my life at ease or will I have a constant burden. To end I will say this... It doesn't matter if you know too much, too little, or nothing at all; as long as you're happy and live life to the fullest it does not matter. So my final answer to this question is It is better to know and not know.
-Jennifer Resendiz 08/27/15 12:13am |
Jennifer R.Grade: 10th Archives
November 2015
Categories |

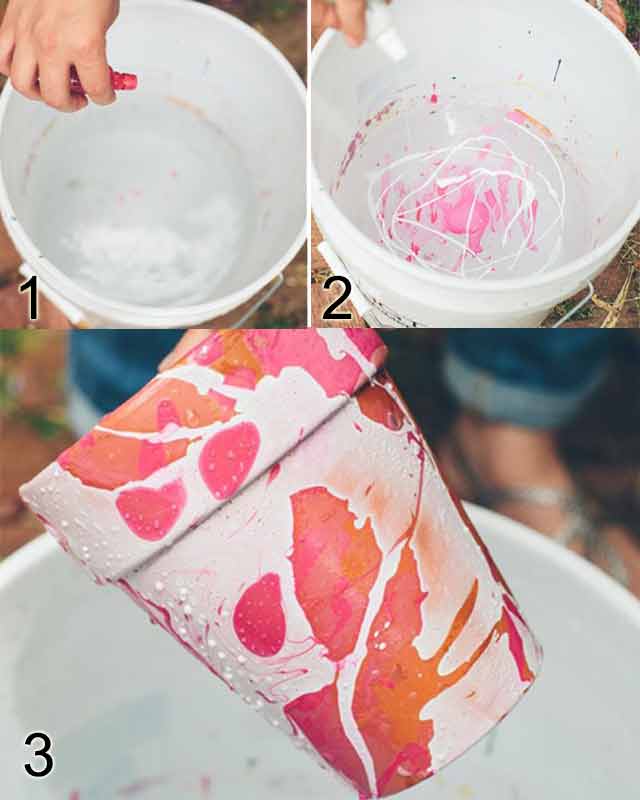

 RSS Feed
RSS Feed
